Welcome to our section on Data Access at HQIP where you can find information about our data access processes (including our Data Access Request Group, DARG), and how we provide safe access to data.
If you are a prospective data applicant and would like more information regarding the application process, please head to our information page below:
What do we mean by ‘data access’?
The audits and outcome reviews in the NCAPOP collect, analyse and report data to help improve the quality and outcomes of care in the NHS. We call this the ‘primary use’ of the data. But there may be ways to re-use the data and achieve even more public benefit – for example for other healthcare improvement initiatives, projects, service planning or innovation projects. We call this ‘secondary use’ of the data, and we use the term ‘data access’ to describe how we review applications for secondary use projects, provide permission and then share or allow access to the data for these projects.
HQIP’s Data Access Request Group
HQIP’s Data Access Request Group (DARG) is the committee that is responsible for considering applications for ‘secondary use’ of data and recommending to HQIP whether the proposed access to data should be supported. It is important that DARG ensures that any data access agreed is anticipated to benefit the health and care of the public. It is also essential to ensure that the data will be used appropriately, safely, and legally (see the Five Safes Framework section below).
The data access request process – how does it work?
An individual, or organisation, that wishes to request access to data must submit an application (known as the Data Access Request Form) to HQIP’s DARG. HQIP’s data access process has two distinct phases: approval and dissemination.
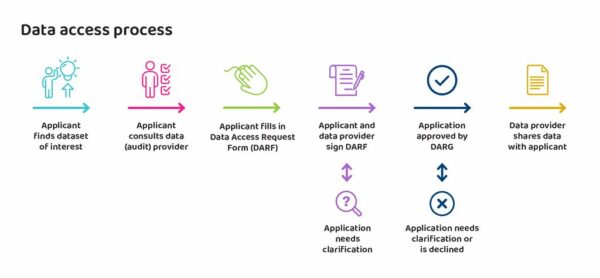
Firstly, to gain approval, the application is reviewed by HQIP and by the audit provider that is commissioned by HQIP to run the relevant NCAPOP project. The purpose of the review is to determine whether the requested data can and should be shared. The application is reviewed first by the NCAPOP provider who checks that the proposed use is clear, robust, achievable, and likely to result in the anticipated benefits. If so, they sign the application which is then reviewed by HQIP at DARG.
DARG then systematically reviews the application at one of their monthly meetings. There are often clarifications and extra information requested, and sometimes applications are not successful.
If the application is approved, HQIP adds their signature to the application form which the NCAPOP project and applicant have already signed, and which also includes the terms of the data sharing agreement. Once this is in place, the second phase, dissemination, can follow. In this phase the data detailed in the agreement is prepared by the NCAPOP project team and shared with the applicant. Whilst HQIP’s DARG manage the data access request process, the actual data is held and released by the NCAPOP team.
Understanding health data and access
Complex rules govern how health and care data can be shared for research, innovation, and other uses beyond individual patient care. Making sense of, and keeping up to date with, how these systems work to provide access to data can be daunting, both for new data applicants and for patients and the public. Our Understanding Health Data Access webpage provides some informative resources that outline how to design projects which protect patient privacy and meet legal requirements. Included in this is a section dedicated to providing information for patients and the public, with useful links signposting to other resources available for individuals who would like to find out more. You can also find out more about how patient data is used and why it’s important through the resources created by Understanding Patient Data.
The Five Safes framework – how do we provide safe access to data?
The Five Safes framework is a set of principles, originating from the Office of National Statistics (ONS), that enable organisations to ensure they are providing safe access to data. It provides a way of thinking through the checks needed to be sure that the data will be used in a secure and responsible way. The Five Safes are: safe people, safe projects, safe settings, safe data, and safe outputs.
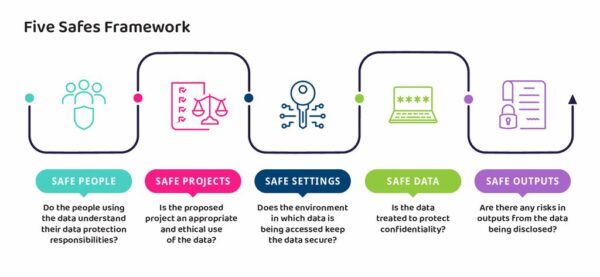
The Five Safes are used by many organisations to support the safe use of data – more information regarding the framework can be found on the UK Data Service’s webpage and their introductory video.
Copyright requests
If you would like to use materials such as tables, text and other information, from the programmes we commission and host, then see the Copyrights requests section of the Policies page on this site.