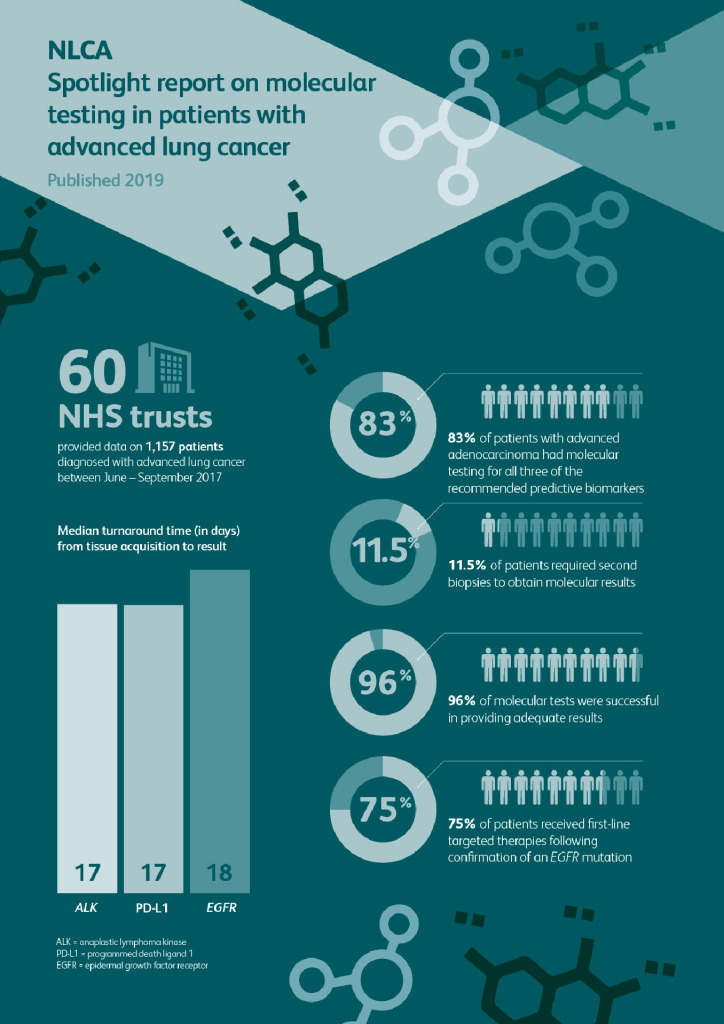
This spotlight audit is the first national study investigating the efficacy and outcomes of molecular testing in patients with advanced non-small-cell lung cancer (NSCLC) in England. In total 60 trusts provided data on 1,157 patients.
Some of the key findings include:
- 83% of patients with advanced adenocarcinoma underwent molecular testing for all three of the recommended predictive biomarkers (EGFR, ALK and PD-L1).
- 96% of molecular tests were successful in providing adequate results.
- 11.5% of patients required second biopsies to obtain molecular results. There was an increased likelihood of patients requiring a second sampling technique if the initial attempt was through pleural biopsy or aspiration.
- The median turnaround time from tissue acquisition to EGFR mutation result was 18 days.
- First-line targeted therapies were received by 75% of patients following confirmation of an EGFR mutation and 58% of patients with an ALK translocation.